|
3/18/2024 0 Comments Bohrs model of an atom 
The circular orbit labelled "n=1" closest to the nucleus has the smallest radius, and therefore the lowest energy. Radius of the orbit increases as you move away from the nucleus. The small positively charged nucleus is shown as a point at the center of the diagram.Įach circle represents a possible circular orbit (stationary state) for an electron.Įach circular orbit (stationary state) has a particular energy and therefore a particular radius.Įnergy increases as you move away from the nucleus. The first five circular Bohr orbits or stationary states are shown on the right. 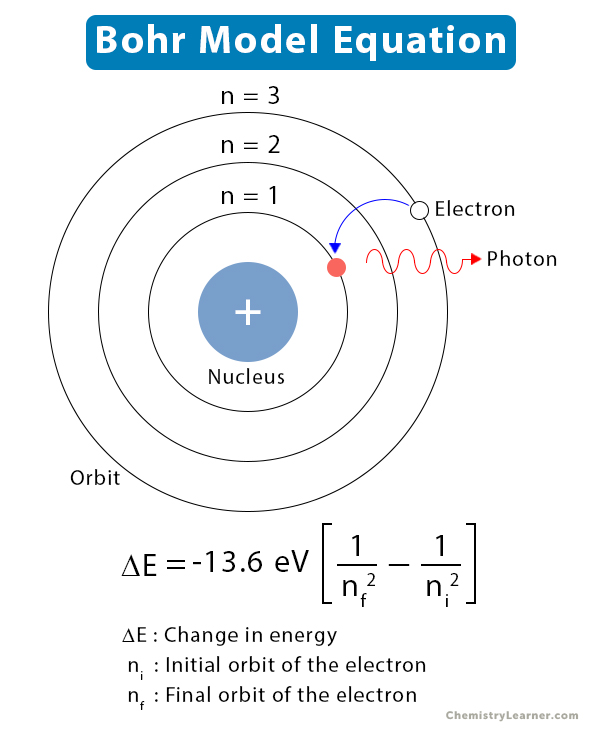
An electron in this stationary state does not radiate energy. 
The Rutherford model predicted an unstable atom, one in which the negatively charged electron spiralled into the positively charged nucleus, emitting energy continuously.īohr overcame this difficulty by assuming a quantum description for energy transfer, and by stating that classical theory did not apply because atoms were stable.Įlectrons could only exist in particular fixed energy states which he called stationary states. Niels Bohr applied the quantum concept of quantisation of energy, energy can only exist in discrete packets of energy, to Rutherford's classical model in order to explain the stability of the hydrogen atom and its discrete emission spectrum. Play the game now! The Bohr Model of the Atom The frequency of the light emitted should be changing continuously.Īn element's emission spectrum would be a continuous spectrum like that shown below:.An electron moving like this should be continually emitting light, losing energy, and spiralling into the nucleus.Īccording to the Rutherford model an atom should be unstable, but atoms are observed to be stable.Electrons occupy the rest of the space, moving rapidly in circular orbits around the nucleus.Ĭoulomb attraction between negatively charged electrons and positively charged nucleus being just sufficient to supply the necessary centripetal force to keep the electrons in orbit.Nearly all the mass and the positive charge is concentrated in a small central nucleus.No ads = no money for us = no free stuff for you! Models of the Atom Before BohrĮrnest Rutherford had proposed a nuclear model for the atom before Niels Bohr. relative intensities of spectral lines in a spectrum.spectra of atoms or ions with more than 1 electron.Limitations of the Bohr model of the atom. The electron can only exist in certain discrete energy levels, therefore only particular orbits are available to it.If the electron falls back to an orbit with a smaller radius, one of lower energy, then the excess energy is emitted.If an electron absorbs energy it moves to an orbit with a larger radius, that is, an orbit of greater energy.Electrons moved around the nucleus in fixed orbits.Īn electron in a particular orbit has a constant amount of energy.In 1913 Niels Bohr proposed a model for atomic structure based on the following postulates: You need to become an AUS-e-TUTE Member! Bohr Model of the Atom Chemistry Tutorial Key Concepts Want chemistry games, drills, tests and more? Bohr Model of Atomic Structure Chemistry Tutorial More Free Tutorials Become a Member Members Log‐in Contact Us 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
